Genetic modification was not invented in 2015. DNA was edited before CRISPR/Cas 9, just as books were printed before the Gutenberg Bible. Is it crazy to compare CRISPR to the printing press? Perhaps, time will tell. But the comparison does illustrate the enormous transformative power of technology made cheaper, faster and more efficient. It is hard to overstate the likely impact of CRISPR on medicine; it is already revolutionizing the development of new therapeutics from gene therapy to stem cell therapy to customized cell lines for drug development. Improvements to the technology and new applications for use have come so thick and fast that at times it seems like #crisprfacts, the hashtag invented to mock the CRISPR hype, can hardly keep up.



Here’s mine…
Now is the winter of our discontent made glorious summer by CRISPR. #crisprfacts
Oh, yeah, and some other things happened too. Here’s the countdown:
- Roche Buys Billion Dollar Stake in Foundation Medicine
In January 2015, the Swiss pharmaceutical company Roche spent just over 1 billion dollars to obtain a majority stake in Foundation Medicine, a pioneer in cancer genomic testing. The deal not only symbolizes but may catalyze the mainstream role of genomics in cancer therapy, as tumor testing continues its rapid ascent from cameo performer to standard of care.
Foundation, which has yet to turn a profit, offers separate tests for solid tumors and blood-based malignancies. The tests offer sequencing of a large number of genes known to be implicated in cancer, but fall short of exome sequencing and examine only cancerous cells and not the germline comparison. Foundation reports are intended to help oncologists choose therapeutic options, including drugs and clinical trials. Roche’s involvement should increase marketing of the tests in the U.S. and abroad, and they likely hope that it will bolster research, such as identifying the markers of tumor DNA that could provide the basis for the highly anticipated ‘liquid biopsies’.
- Matchmaker Exchange Goes Live

When you’re driving in traffic, other people are annoying. When you are in line at the supermarket, other people are annoying. But when you are trying to solve medical mysteries with a genetic test, other people are the answer.
Parenting a child with an undiagnosed genetic disease is a trip without guidebooks. Treatment is a series of guesses, prognosis is unknown. No one can warn you about what’s to come, or reassure you about what will pass. Genetic testing may reveal the apparent cause, but in cases where the variant has not been seen before it can only be confirmed by the second case. Patient networks built around genotype can improve treatment, clarify reproductive risk and provide emotional support.
Because clinically significant genetic changes are individually rare and collectively common, finding another person with the same gene variant or the same mutation in a tumor requires access to vast amounts of information and the means of searching it. Fortunately for us, we live in an age defined by the ability to access vast amounts of information and the means search it. But sharing genetic information on the internet has been complicated by rules designed to protect patient privacy and the hot mess that is our patient records system.
In September, a team led by Heidi Rehm announced the launch of the Matchmaker Exchange, a collaboration with multiple partners that provides secure sharing of patient information linking phenotype and genotype. Rehm described the new venture as “a reliable, scalable way to find matching cases and identify their genetic causes.” Congratulations to the field of genomics, and welcome to the Internet Age.
- Illumina Launches Helix, a Consumer Genomics Platform

In 2015, the consumer genomics industry is not so much an industry as it is a high tech field of dreams, a plowed-under cornfield in the cloud, waiting for the crowds to arrive. “They will come,” says the prophet in the James Earl Jones voiceover voice, “not even knowing for sure why they’re doing it. They’ll arrive at your door as innocent as children, longing for the future. They will pass over the money without even thinking about it; for it is money they have and peace they lack.”
But while back in Iowa poor Ray had to fight the bankers to keep his dream of a self-sustaining ghost baseball industry alive, capitalists are lining up to host the field of genomes. Both Google and Apple have cloud-based storage systems for DNA sequence data; Illumina’s proposal is unique in that you pay not for storage but for use. The company is betting that multiple third parties will develop consumer applications that require genomic information, smartphone apps that personalize your risk for side effects from pharmaceuticals or calculate the degree of relationship between you and your Tinder match. Helix holds onto your genomic digits the way Amazon holds onto your credit card information, making it easier for each new purchase to flow through them.
Illumina, the undisputed heavyweight champion of second generation sequencing, makes a forward-looking move here, tilling the soil in a hypothetical ecosystem. Two years ago, the ‘consumer genomics industry’ was a fancy synonym for 23andMe, one single tree that dominated the landscape. Ironically, the FDA pruning of 23andMe in 2013 that cut back their health and wellness business provided a little sunshine for smaller farmers, and in 2015 the first green leaves of a thousand consumer genomics products popped up out of the dirt, offering gene-based advice on the treatment of mental illness, on diets to suit your metabolic type, on the probability of cardiac events. These new shoots are individually weak – in many cases not rooted in the science, in others likely to be mown down by regulatory mechanisms not yet in place – but collectively they represent a widespread belief that there is money to be made in these fields.
- In Memento Moratorium
“It is easier to stay out than to get out.”
–Mark Twain
On April 18th, a group of Chinese scientists led by Junjio Huang published a paper in Protein and Cell describing their attempt to edit (but not implant) human embryos using the CRISPR/Cas 9 system. The goal was to alter the hemoglobin-B gene, which happened in 4 out of 54 embryos, although all 4 were mosaic – some cells were altered and others were not. This, the authors concluded, was not a success. Improving “fidelity and specificity,” they wrote, is a “prerequisite for any clinical applications of CRISPR/Cas 9-mediated editing.”
But failure or no, the publication ignited a firestorm of debate. On one thing the scientific community agreed: the experiment was evidence that the question of to edit or not to edit is in the offing. Improvements in the efficiency of gene editing are occurring so fast that the technology used in the study was itself a generation or so out of date before it made it into print. Can we do this? Not yet, say the authors of this paper. Should we do this? That is a much harder question, a question that launched a thousand editorials in 2015.
Early debates about what should or should not be allowed in DNA engineering did not focus on the human germline, but the consensus that evolved drew a line between somatic human uses for gene therapy, and changes that would affect eggs, sperm or embryos. Avoiding changes that would be passed down through generations confined any unintended effects to the individual, and sidestepped all the societal issues wrapped up in the concept of ‘designer babies.’ The moratorium that some scientists called for after word spread of the beta thal experiment is not new, and if heeded would reinstate a tacit agreement that had been in place since the 1970’s.
Oh, but it is easy to say you wouldn’t do something when you can’t. The Chinese paper resulted in an international summit on human gene editing in December, hosted by the National Academy of Sciences. The statement produced after 3 days of meetings endorsed somatic uses and germline research, but labeled any clinical use (i.e., use that could result in a baby with edited genes) irresponsible – for now. The note of caution may have obscured what is effectively a rejection of any hard and fast limitations. “As scientific knowledge advances and societal views evolve,” the organizers wrote, “the clinical use of germline editing should be revisited on a regular basis.”
- Sequenom Introduces a Non-Invasive Scan of the Genome
“Facts are stubborn, but statistics are more pliable.”
–Mark Twain
In September 2015, Sequenom launched MaterniT Genome, an expanded version of its non-invasive prenatal screen designed to catch all microdeletions or duplications greater than or equal to 7 MB. This is simultaneously not that important at all and an illustration of everything we are dealing with now and a window into the future.
The new Sequenom test joins its stablemates VisiblitiT (tests for trisomies 21 and 18) and MaterniT Plus (tests for all the trisomies plus select, well-characterized microdeletion syndromes like Wolf-Hirschorn or Cri-du-chat). All the tests report on fetal sex. Everybody reports on sex, and the most common form of informed consent for testing consists of an obstetrician asking the patient “do you want to do the test for gender?” (I can’t prove this but it’s true. Ask around.).
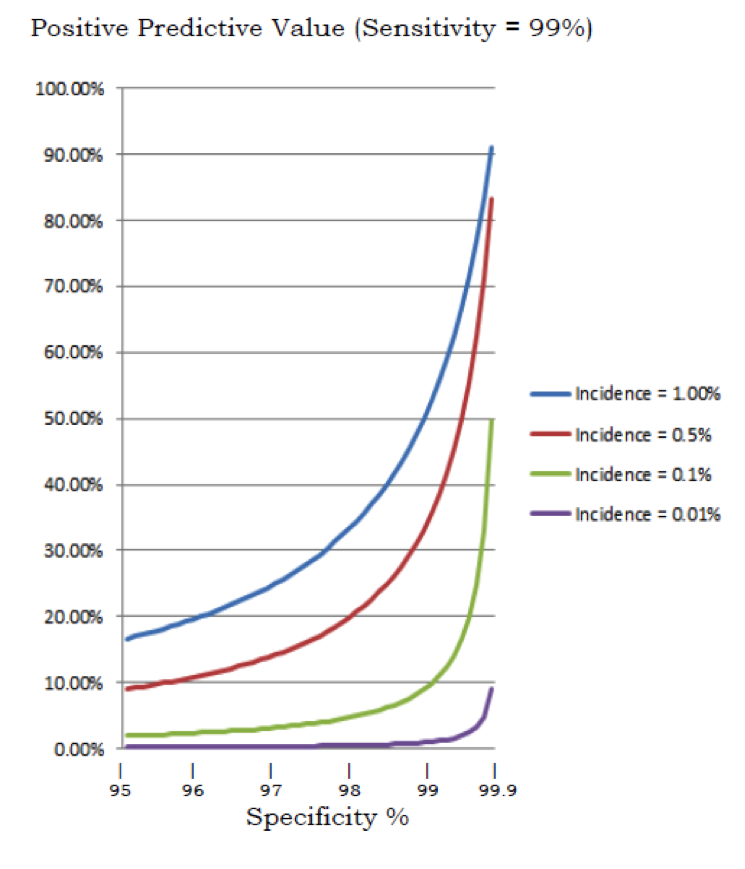
Of the three other U.S. purveyors of non-invasive testing, only Natera includes the option of a microdeletion panel. Although NIPT is the hottest selling thing in the universe, reaction to the microdeletion panels have been lukewarm, and here’s why: math. The Achilles heel of NIPT is positive predictive value, or the percent of the time that the test flags a pregnancy and is wrong. Even when a test is very accurate, the rarer the condition, the higher the percentage of false positives. Doctors and genetic counselors don’t like false positives because in real life a ‘false positive’ is a very frightened and very upset patient, and in real life some of these patients have ignored advice for follow up and terminated pregnancies that turned out to be unaffected (this sounds very extreme but remember that they are looking at a test labeled 99+% accurate, and under intense time pressure at just around the point when most people go public with a pregnancy).
Microdeletion syndromes are rarer than trisomies, so even as accuracy remains high, positive predictive value drops precipitously. Sequenom offers no estimates of PPV, and Natera’s own numbers suggest a PPV of just 5.3% for 22q11 deletion syndrome. In this context, the Sequenom genome-wide test seems like a curious step. Not only does it raise serious questions about PPV, but most of the deletions and duplications would be uncharacterized, meaning that counseling patients on the predicted effect of the change would be complex. None of this is exactly obvious in the Sequenom promotional material, which highlights 99.9% specificity and 92.9% sensitivity.
Why is a test likely to be used sparingly a top story for 2015? Because it has a ‘more information/less clarity’ aspect that is very 2015. Because it shows the quandaries into which we wander, when we take our limited 2015 knowledge into the realm of prenatal testing. And… because limited use may grow over time, as Sequenom no doubt knows, so that this may well be a first look at the prenatal testing of the future.
- Gene Expression? There’s a CRISPR for that.

When exactly did the reports on CRISPR start to sound like an infomercial? Maybe it was March of 2015, when scientists from Duke University led by Timothy Reddy and Charles Gersbach published an article describing their success using an adapted CRISPR/Cas 9 system to create a targeted increase in gene production.
CRISPR! It slices, it dices… No wait, there’s more…
In this case, the modified CRISPR program links a guide RNA that searches out the target DNA with a protein that catalyzes acetylation – so instead of gripping and snipping, your bonus CRISPR tool finds the appointed enhancer region and flips a switch, turning gene production on. And voila: “A programmable, CRISPR-Cas9-based acetyltransferase…leading to robust transcriptional activation of target genes from promoters and both proximal and distal enhancers.”
Clap on, clap off… the Clapper!
Debates may yet rage about the nature of epigenetics and its intergenerational significance (hell, spellcheck still refuses to recognize it as a word) but no one argues about the importance of gene expression. Changes in gene expression are central to both development and stasis; altering gene expression provides a possible avenue of control of every process from learning to aging.
Amazing! And for far less than you might think! Does it come in red?
- A Prenatal Genetic Test Reveals Cancer in the Mom-to-Be
In the four years since non-invasive prenatal testing was introduced it has grown into a market worth over half a billion dollars annually in the US alone, with double digit growth projected for years to come. The number of invasive procedures has fallen off a cliff, with many women opting not to do amniocentesis or CVS after reassuring results on a non-invasive prenatal screen. But not everyone has been reassured. In March of this year, Virginia Hughes at Buzzfeed reported on the case of Eunice Lee, who learned she had cancer after the lab reported unusual results on her non-invasive screen.
This rare event – Sequenom suggested that one in 100,000 of their tests results pointed at a malignancy, with just over half of those subsequently confirmed – affects only the (thankfully) limited universe of pregnant women with cancer, but the story is more universally significant for at least two reasons.
The first is how it reflects the challenges surrounding non-invasive testing, the first major testing modality to roll out as an industry unto itself. Since it’s inception, this technology has developed in a highly competitive and market-oriented environment (one Sequenom executive lied about early test results and would have gone to jail if she hadn’t died first) and many people have suggested that their pre-market studies were inadequate and self-serving. The FDA has pointed to non-invasive testing as an example of why laboratory-developed tests need more regulation. All of this criticism has continued despite the fact that the tests are extremely popular and largely successful, and have decreased the need for more expensive and more dangerous invasive testing. Because it is so new and because the early studies were limited, these funky results are an anomaly that put the testing company into an awkward spot. Although they look like cancer, they can’t be officially reported as cancer, because there are no studies to validate that claim. Ignoring them, on the other hand, seems like an ethical breach to me, given that there is some evidence that suspicions are correct. Sequenom chose to call the test non-informative, but alert the physician to their hunch. Other companies have chosen to say nothing in similar circumstances.
The second take home point of this story is how close we are to a new type of cancer diagnostic, one that will be used both as a screen and a test for recurrence or the effectiveness of chemotherapy. If prenatal testing is any model (and it is) it will appear soon, all the companies involved will sue one another frequently, and we will all work out the bumps as we go along. One of these days we will all be surprised to read about someone concerned about cancer who discovered she was pregnant.

Eunice and Benjamin Lee
Ms. Lee, by the way, was successfully treated for colon cancer with surgery alone, and gave birth to Benjamin, a healthy baby boy.
- Baby With Cancer Responds to Treatment Using Genetically Modified Cells
The headline for this segment should have been, First Clinical Use of CRISPR Technology Saves Baby With Cancer, except no part of that sentence is true. The gene modification technology used wasn’t CRISPR but Talens, an older approach that is more expensive, less flexible and more technically demanding. It wasn’t the first use of gene modification as a therapy, just the first that presents a promising path to widespread use. And let’s not jinx the baby, five months into remission, with an overconfident use of the word cured.
And yet, 18-month-old Layla Richards is home with her dad and mom (probably mum; they say mum in Britain) 6 months after doctors counseled the family to consider palliative care for acute lymphoblastic anemia. If there was a miracle involved, it was simply the miracle of being in the right time and the right place – Great Ormond Street Hospital in London, which had on hand modified T cells intended for use in a clinical trial for the French biotech company Cellectis, slated to begin in 2016. The Cellectis process involves knocking out a gene in donor T cells so that they cannot attack host tissues – a step that eliminates the need to use the patient’s own cells, a personalized approach that makes it slower and more expensive. Several companies that have been developing autologous approaches saw their stock prices fall in the wake of this announcement. In the case of baby Layla, doctors say they were unable to find enough T cells to extract for treatment.
Who did what first is a subject best left to the historians (and the patent lawyers). This story represents where we stand in 2015, on the cusp of therapeutic innovation built not on serendipity, the great innovative engine of the past, but on knowledge and engineering. We are entering an age of miracles that are not miracles at all, because we can both explain and reproduce them. And we are entering it fast, with technology out of date before the gun goes off, like thoroughbreds groomed and trained who show up at the starting gate to find themselves racing unicorns.
- First Analysis of Large Data Sets Suggests: When It Comes to Variant Classification, It’s Clinician Beware, At Least For Now
“The trouble with the world is not that people know too little; it’s that they know so many things that just aren’t so.”
-Mark Twain
Anyone arrogant enough to believe we were equipped to interpret the human genome must have found the last few years humbling, poor foolish person. But most of us, veterans of the diagnostic odyssey and the variant of uncertain significance, were prepared to admit that it was early days. The collective need for more information has in recent years overcome proprietary and competitive instincts, and convinced many researchers and commercial laboratories to share their data. The top story for 2014 was ExAC, a Broad Institute initiative that has aggregated exome data from over 60,000 healthy adults.
Preliminary analysis of that data is in, with a couple of headlines. One – no surprise – there’s a lot we don’t know. As expected, mutations that result in a loss of function are constrained in genes associated with severe disease – in healthy individuals, you should see limited loss of function in genes where disruption causes a severe phenotype. We saw this purifying effect in many genes, and 79% of them are not yet associated with human disease. That’s the knowledge gap that we need to fill.
Headline number two: lots of things we thought we knew are wrong. The extent of this may qualify as a surprise, although careful observers will not be shocked. Plenty of evidence existed that existing databases and analyses were larded with inaccuracies. The database ClinGen reported in June that among the 12,895 unique variants with clinical interpretation from more than one source, 17% were interpreted differently by the submitters. The ACMG guidelines for variant interpretations published in March stressed that variant “analysis is, at present, imperfect, and the variant category reported does not imply 100% certainty.” Analysis of ExAC, a preliminary report suggests, shows that most individuals carry a rare and presumably deleterious variant in a gene associated with dominant disease. Beyond inaccurate classification, this may be evidence of incomplete penetrance, subclinical presentations, or simply the resilience of the genome. Take home point, as stated by Dan MacArthur et al, “The abundance of rare functional variation in many disease genes in ExAC is a reminder that such variants should not be assumed to be causal or highly penetrant with careful segregation or case-control analysis.”
- The Power of Gene Drive
“The only difference between reality and fiction, is that fiction needs to be credible.” –Mark Twain
Do you know that moment in the movie when the hero has to decide whether or not to commit some morally ambiguous act in order to save thousands of lives? Remember that? Well, forget about it. That make-believe drama cannot compare with the real life dilemma facing scientists, regulators — all of us, actually – in light of this year’s signature story, a CRISPR-mediated system that can rewrite the laws of evolution to propagate traits devised in the laboratory.
Gene drive is a term for a biological process that increases the probability that a given gene will be passed along to the next generation. In 2014, Kevin Esvelt and George Church at Harvard (et al) wrote a paper describing how CRISPR could be used to insert a tricked-out version of an edited gene that included the machinery to hack out the corresponding gene from the other parent and replace it with a copy of itself, complete with the gene drive complex. Introduce this zombie gene into any fast-replicating population and the allele frequency doubles with each new generation until there aren’t so many wildtype alleles left to convert.
Welcome to 2015, when a hypothetical is always just one grad student project away from reality. In November, Sharon Begley at STAT reported that success with fruit flies in the UC San Diego lab of Ethan Bier had led to a collaboration with UC Irvine’s Anthony James, who has developed an edited mosquito gene that destroys the parasite that causes malaria. Success could mean the most effective means of malarial control ever devised, and one that effectively spreads itself.
Herein lies the dilemma: this intervention is not so much introduced as unleashed. Although Church and Esvelt recently published a paper detailing strategies for containment and reversal of gene drives, concerns remain over the specter of unintended consequences. The Pentagon and the United Nations are reported to be concerned about the potential for weaponized insects. Scientists and ethicists have expressed alarm about the unknowns associated with any disruption of an evolved ecosystem. But the WHO reports that in 2015 there were 214 million cases of malaria and almost half a million deaths. So here’s the movie pitch: the mosquito is a terrorist killing 1500 children every day. You, the scientist, can reprogram the mosquito, with unknown impact on the entire planet. The developing and developed world can’t overcome their mutual distrust to make a plan. Do you release the zombie mosquito?
Buy it as a movie? No one would. It’s just too out there.

Follow me on Twitter