Things they do look awful c-c-cold/Why don’t you all f-fade away?/And don’t try to d-dig what we all s-s-say/Just talkin’ ’bout my g-g-generation – “My Generation” by Pete Townshend and The Who
The boast in the title of this piece is not aimed at the generation or two of genetic counselors who entered the field after I graduated from the UC Irvine genetic counseling program in 1983. Instead, it verbalizes the ethical conceit on the part of genetic counselors and medical geneticists in any given era that previous generations of geneticists always seem to have glaring ethical lapses. Yes sirree Bob, we learned our historical lessons from studying what they did wrong. We are good people with high ethical standards and would never say or do anything as bad as they did. Our moral compass is pointed to True Ethical North.
And so we create self-serving historical narratives that tell a tale of past sins and generational redemption.* Like Dante’s Divine Comedy, the genetic counseling narrative is a journey from Inferno (classical eugenics) to Purgatorio (post WW-II and the professionalization of medical genetics) to Paradiso (nowadays). Each step of the journey we strive to become better people and better genetic counselors, though the comedy may not be so divine. Think of me as your Virgil, and follow me on a journey through the history of genetic counseling and medical genetics.
![The Barque of Dante (French: La Barque de Dante), also Dante and Virgil in Hell (Dante et Virgile aux enfers), is the first major painting by the French artist Eugène Delacroix, and is a work signalling the shift in the character of narrative painting, from Neo-Classicism towards Romanticism.[1] The painting loosely depicts events narrated in canto eight of Dante's Inferno; a leaden, smoky mist and the blazing City of Dis form the backdrop against which the poet Dante fearfully endures his crossing of the River Styx. As his barque ploughs through waters heaving with tormented souls, Dante is steadied by Virgil, the learned poet of Classical antiquity.
Pictorially, the arrangement of a group of central, upright figures, and the rational arrangement of subsidiary figures in studied poses, all in horizontal planes, complies with the tenets of the cool and reflective Neo-Classicism that had dominated French painting for nearly four decades. The Barque of Dante was completed for the opening of the Salon of 1822, and currently hangs in the Musée du Louvre, Paris](https://thednaexchange.com/wp-content/uploads/2026/04/image-13.png?w=960)
Inferno
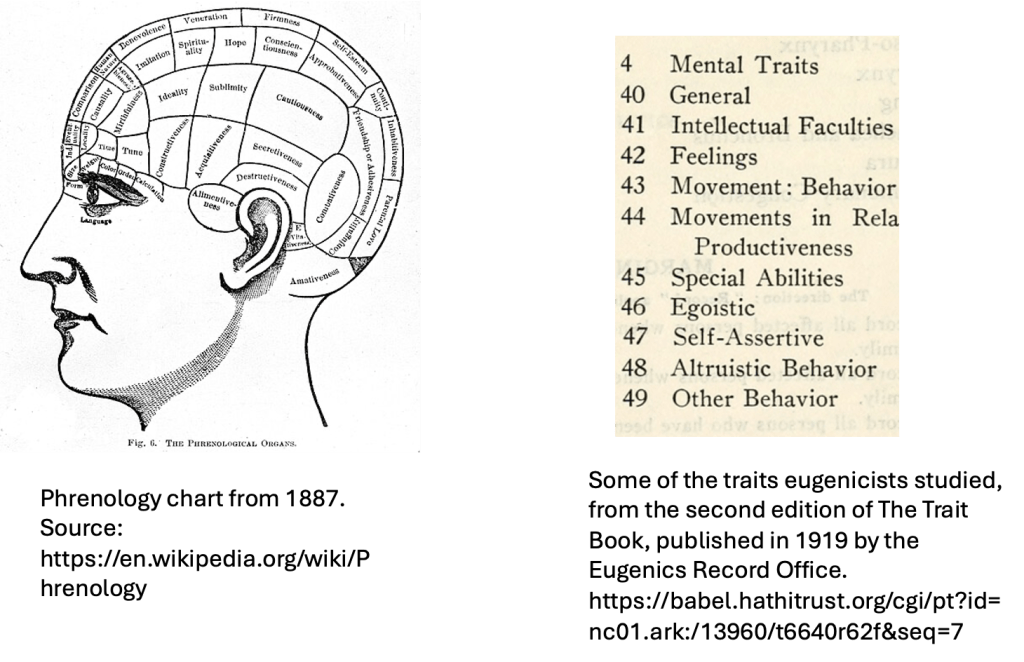
The scientific study of human genetics was conceived with the Original Sin of Eugenics on its soul (keeping up Dante’s religious imagery). For several decades into the 20th century, genetics and eugenics were more or less inseparable. Almost all major geneticists espoused eugenic ideology. And no, eugenics was not a pseudoscience; that is another conceit of the present passing judgment on the knowledge base of the past. In fact, eugenicists themselves dismissed its predictive forebear, phrenology, as a pseudoscience while heralding genetics as a true science of human traits and behavior. But to our 21st century perception, there is a slim difference between reading skulls and reading pedigrees.
Eugenicallly-minded geneticists were not stupid; many were brilliant scientists who had a very sophisticated understanding of genetics, the environment, and statistics, at least within the context of the times. Highly respected geneticists such as Karl Pearson, Herman Muller, Lancelot Hogben, Ronald A. Fisher, and Raymond Pearl criticized some of the less sophisticated eugenic applications of genetic knowledge but nonetheless remained staunch eugenicists. Heck, even Franz Boas, the anthropologist who is often cited as the ultimate anti-eugenicist, supported sterilization for reasons of “hereditary unfitness.” Boas, one of the most forceful critics of theories of racial superiority, also internalized some of the standard beliefs of the time about so-called primitive cultures. In 1897, he had the polar explorer Robert Peary essentially kidnap six Greenland Inuqhuit men, women, and children and bring them to the American Museum of Natural History in New York City so they could be studied as biological and anthropological specimens.**
I am not trying to justify eugenic ideology or programs or serve as an apologist. There were many different versions of eugenics that arose around the globe and over time (and continue to do so). Each version was bad, but often bad in its own way. Eugenics was pervasive and cut across all economic and ethnic segments of society. It was taught in high schools and colleges, it appeared in advertisements for consumer products, and newspaper stories. Support of eugenics wasn’t universal but it sure was widespread.

The biases and beliefs of earlhy 20th century geneticists reflected some of the major social issues of their day – large scale immigration from non-Western European countries of people thought to be morally, intellectually, and physically inferior; the growth and increasing visibility of urban poverty with increasing urbanization along side an extremely wealthy upper class; the supposedly low IQ and poor health of the millions of soldiers who enlisted or were drafted to fight in the First World War (and English soldiers who fought in the Boer War). Geneticists from this period were products of these times who also simultaneously helped shape their era. It is also unsurprising that the vast majority of the early eugenicists were men at a time when men still felt that they had the right to make reproductive, economic, and other life decisions for women. It is easy to call out their moral shortcomings – bigotry, racism, smug moral superiority, ableism, classism, sexism, etc. Some eugenicists were truly evil and many inflicted all kinds of awful harm but for the most part they were a bunch of flawed human beings, just like we are. They were reacting to what they perceived to be threats to their way of life and to (White Western European-centric) civilization, and many thought eugenics was a way of helping eugenically inferior people.

Purgatorio
The next phase in the historical development of genetic counseling was the rise of medical genetics as a clinical and research discipline in the decades following the end of the Second World War, critically examined in Nathaniel Comfort’s classic book The Science of Human Perfection. The mytho-story often told – which, zombie-like, refuses die and that Comfort and other historians have long repudiated – is that the founding figures of medical genetics and genetic counseling specifically rejected eugenic ideology, in part driven by the horrific practices of Nazi Germany. Medical geneticists took great pains to distance themselves from the excesses of their eugenicist forebears and criticized eugenics for its ethical shortcomings and simplistic understanding of genetics. Indeed, in 1947 Sheldon Reed famously defined genetic counseling as “a kind of genetic social work without eugenic connotations.” But, at the same time, many geneticists maintained membership in the American Eugenics Society and espoused beliefs and genetic counseling principles that to 21st century sensibilities sounds an awful lot like eugenics disguised as “educated parental choice” (Reed joined the society in 1956 and remained a member through nearly 2 decades and a few name changes of the society).

Some of the major social issues of this period were the worry about over-population of the earth (largely in poorer, non-White countries) in the context of supposedly limited resources, the health and genetic effects of ionizing radiation from nuclear bomb testing and detonation sparked by the detonation of atomic bombs over Nagasaki and Hiroshima and the ensuing nuclear arms race between the United States and Russia, the alleged relaxation of evolutionary selection due to advances in medical care and social changes that allowed survival of people who were genetically predisposed to medical conditions who might ordinarily have had no or few offspring, and the birth of babies with congenital conditions due to fetal exposure to rubella and thalidomide. Geneticists expressed their concerns for the quality of the human gene pool and the health of babies in the context of these issues and recommended various measures to ensure the genetic health of the population, such as greater access to contraception, reducing population growth, limiting exposure to ionizing radiation, and genetic testing and counseling. All while trying to distance themselves from eugenics.
Trouble in Paradiso
In the next phase of the journey, during the 1970s, the profession of genetic counseling was established in the United States, with the founding of the first masters level genetic counseling programs at Rutgers University, the University of California Irvine, the University of California Berkeley, and Sarah Lawrence College, among others. From its inception – and up to today – the profession was overwhelmingly female, in stark contrast to previous generations of medical geneticists who, nearly to a man, were, well, men. Most of the first professional genetic counselors – and many of their patients – were middle and upper middle class, highly educated, politically liberal women who were aligned with the key issues of Second Wave Feminism, particularly support of abortion, reproductive rights, and rejection of patriarchal authority in medicine. For several decades, publications by genetic counselors, and their professional Code of Ethics, focused on a feminist ethic particularly in reproductive matters and explicitly rejected eugenic ideology (although they were not exempt from eugenic criticism, especially from disability and feminist scholars). The demographic profile of genetic counselors remained largely unchanged for several decades.
The profession sometimes self-critically recognized its homogeneity but did not make concerted efforts to change the historical demographic profile. It was only over the last 5-10 years or so that more people have entered the profession who are non-White, of non-European ancestry, openly queer, live with a disability, are neurodiverse, and are male, even if the profession is still skewed toward its historical demographic. The newer less exclusive demographic has led to guiding ethical principals that focus on diversity, equity, inclusion, and justice within the profession and for the patients that genetic counselors serve. These issues are not unique to genetic counseling and are bitterly playing out in many social, political, and professional arenas.
Despite the increasing professonal diversity, several studies, personal recollections, and commentaries by genetic counselors from different backgrounds attest to the friction felt by both the “Old Guard” and the more diverse younger genetic counselors as these policies are implemented (or not) into training and clinical practice. Implicit biases abound in gatekeepers, supervisors, leading figures, research focus, and conferences. And, yeah, that includes me; far be it from me to cast the first stone. My generation of genetic counselors is very understandably criticized for the class, ethnic, and other biases of its practice, training, and ethos. The criticism hurts but probably because its core of truth makes us uncomfortable.
But at some point in the future, a fresh new generation of counselors will level a critique at the current practice, profession, and ethos of genetic counselors. and its focus on diversity, equity, inclusion, and justice. It is impossible to know what they will continue to embrace or decide to reject, or they might wonder how we could support certain viewpoints and practices, or shame us for some things, or criticize the language that we use. They will likely have a whole new set of issues that they feel will need to be freed from past baggage. Or maybe they will think the current generation is the apotheosis of moral practice (though I doubt it). As the history of medical genetics and genetic counseling illustrates, the ethos, morals, and clinical practice are always a microcosm of the larger socio-cultural environment. We are all situated within our eras and our ethnic and socio-economic context.
Though if we can only learn one constant about the history of genetics. it is that eugenics never goes away and it is always bad, whateve form it takes. We have to call it out when we see it, wherever it is. Maybe even in our own practice.
So let us practice ethical humility and recognize that even (especially?) highly ethical people have ethical shortcomings, difficult to admit to and often not obvious to us in the present but glaringly obvious with historical hindsight. We are all trying to do the right thing, even if we can’t all agree on what the right thing is. And the right thing changes over time, and we sometimes can’t appreciate when we are not doing the right thing. We are always on a journey, searching for ethical clarity.
E quindi uscimmo a riveder le stelle.***

_______________________________________________________
*- The historical narrative is broadly similar in the United States, Canada, England and Australia. The history of genetic counseling in the rest of the globe is less well-studied but may not necessarily align with the narrative described here. Nonetheless, I suspect that the central concept of the current generation of genetic counselors passing judgment on the past will be true globally, although the details will differ.
**- Four of the captive Inuqhuit died in New York from acquired illnesses within a few months. The youngest, Minik, a 7 year old boy, was eventually raised as an adopted child of the museum’s chief building superintendent and somehow wound up working as a lumberjack in a small town in New Hampshire, where he died in his late 20s during the influenza epidemic.

*** – The closing line of Dante’s Inferno, often translated as some variation of “And then we emerged to see the stars again.”