This post was modified on 3/30/2022 with input presented by some DNA Exchange readers. First – the original post did not account for the fact that ELP1 is reported out as IKBKAP by Natera, SEMA4 and Quest. Table and graph were updated to reflect this.
While the list of labs surveyed for this post was not intended to include all labs that offer carrier screening, it has been noted since this was initially posted that Fulgent does offer a carrier screening panel based on the ACMG Tier 3 recommendation. A paragraph has been added to reflect Fulgent’s test offering.
In July 2021, the American College of Medical Genetics and Genomics published a new carrier screening guideline, Screening for autosomal recessive and X-linked conditions during pregnancy and preconception: a practice resource of the American College of Medical Genetics and Genomics (ACMG). The gist of this recommendation is that all individuals who are considering pregnancy or who are pregnant should be offered a carrier screening panel inclusive of 113 specific genes. In a prior post, Bob Resta and I shared our concerns regarding ethical issues and the ways in which this panel was designed.
With this post I have a practical question regarding implementation: How does one order a carrier screening panel of these ACMG-recommended genes when such a panel does not exist?
A quick recap of the ACMG carrier recommendation. The guideline defines 4 tiers of carrier screening:

The carrier frequencies utilized are based on the public database gnomAD and represent carrier frequencies for any subpopulation that makes up at least 1% of the United States total population.
The goal of this ACMG recommendation was to define a standard panel that could be offered to all patients. The guideline recommends that all patients who are pregnant or are considering pregnancy be offered the “Tier 3” panel, and that reproductive partners may also be offered the Tier 3 panel simultaneously. This panel is inclusive of 113 genes, 86 of which were derived from the gnomAD data as having a carrier frequency of 1 in 200 or higher; 11 additional genes were “highly represented in one or more patient populations and have potential to be underrepresented in gnomAD”; and 16 genes associated with X-linked conditions with a prevalence of 1 in 40,000 or higher.
ACMG recommends against using the Tier 1 or Tier 2 panel for any patient. Further, it states that the Tier 4 panel be reserved “for consanguineous pregnancies (second cousins or closer) and in couples where family or medical history suggests Tier 4 screening might be beneficial.”
A huge problem with this recommendation is that the Tier 3 panel, as it is described by ACMG, does not exist as an orderable test through most labs. I have surveyed several labs, and those with the biggest commercially available panels do not provide coverage for somewhere between 25 and 49 genes of the 113 that are outlined by ACMG for the Tier 3 panel.
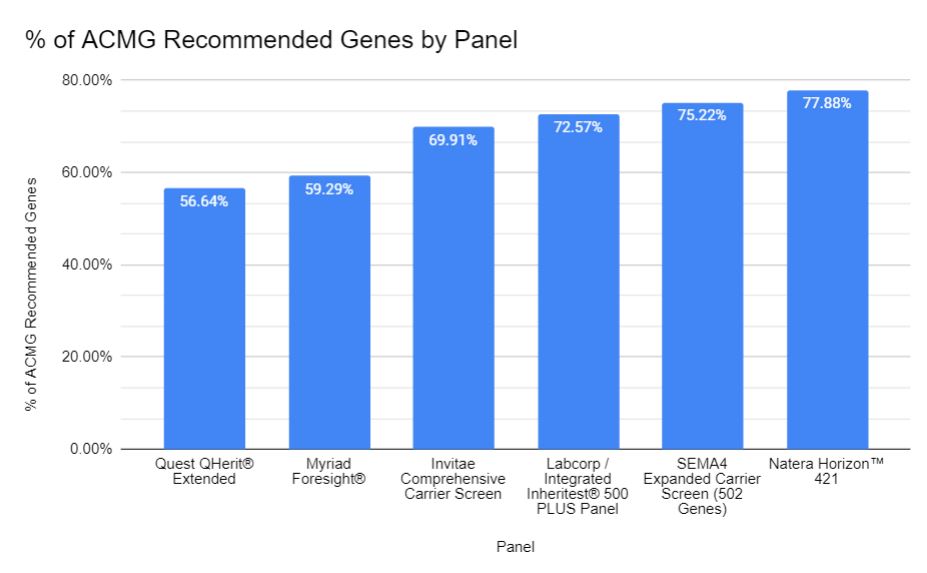
Analysis of carrier screening labs performed on March 24, 2022. For a full list of genes by lab, see Table below.
Some of the labs I have highlighted in this analysis have published press releases and sent emails to their client base in enthusiastic support of these guidelines that call for expanded carrier screening, even though NONE of them actually offer what ACMG currently recommends. The Industry lobbying group for Myriad, Natera, SEMA4 and ThermoFisher, the Access to Equitable Carrier Screening Coalition “applauds American College Of Genetics And Genomics” on this new recommendation that none of these labs can currently fulfill.
One lab, Fulgent Genetics, offers a panel that is based on the Tier 3 recommendation, along with several caveats and limitations for some of the recommended genes that have common variants that may be missed or are expected to escape detection by their platform. While the Fulgent panel includes all 113 ACMG-recommended genes, these caveats and limitations demonstrate that sensitivity across the genes included can vary dramatically and may be low in cases where the common mutation is a trinucleotide repeat, inversion or other alteration not reliably identified through next generation sequencing. For example, Fulgent reports including FXN analysis on their panel with the caveat that their analysis will only detect sequence variants which account for <5% of pathogenic variants in this gene (the vast majority of FXN pathogenic variants are due to GAA repeat expansion.)
Perhaps Fulgent along with other labs that are working to develop a panel that meets the ACMG requirements and will be able to identify most pathogenic variants in the 113 gene set. It may be difficult to do this and keep the cost of testing down as there are technical challenges to assessing some of the genes that ACMG recommends. For example, the most common mutation in the F8 gene is a gene inversion. With PLP1 the most common mutation is a gene duplication. Rearrangements are common with the OCA2 gene. These technical variations present a challenge to labs as they expand their carrier screening panels. From the marketing perspective, adding 50 genes to their currently existing platform may seem more impressive than adding a small handful of more technically difficult ACMG-recommended genes.
Do obstetrical care providers recognize that when they are ordering an expanded panel that it may include hundreds of genes that are not recommended while missing several genes that are part of this ACMG-produced panel? What does this mean for our liability as providers when we cannot order a test that is recommended by our professional society?
In a future post I plan to focus on carrier screening for cystic fibrosis and some of the harm seen from reporting practices on expanded carrier screening. But for now I would like to reflect back to the time when we first considered screening for cystic fibrosis, the rollout of which was not without challenges. Before guidelines were issued by the American College of Obstetricians and Gynecologists and the American College of Medical Genetics, there was significant preparation for implementation. Care was taken to determine whether we knew that the variants included on the CFTR panel cause disease. We thought we were clear about this but even with great scrutiny, the original 25 mutation panel included a variant that we eventually learned did not cause disease. There were standards written for laboratories regarding how to do the testing and what should be included in the report. There were educational resources developed for patients and providers. There appears to be much less care and preparation with these current guidelines in spite of the recommendations for testing for many more as well as increasingly complex conditions.
In today’s world of carrier screening, we see both the 113 gene Tier 3 panel recommended by the ACMG as well as commercial laboratories in constant competition to expand the size of their carrier panels. Yes, labs are expanding their genetic carrier screening offerings, but it does not appear (regardless of their marketing materials) that the recommendations from ACMG are the reason why. Even some of the biggest panels available don’t include 22-43% of the genes recommended by the ACMG while providing coverage for numerous genes that are not included in the recommendation. Data regarding performance characteristics of screening for many of these genes both within and beyond the panel are lacking. As a result, pre and post test counseling our patients regarding carrier screening and the downstream challenges are just going to become increasingly more complex.
ACMG Tier 3 Panel |
Labcorp / Integrated Inheritest® 500 PLUS Panel |
SEMA4 Expanded Carrier Screen (502 Genes) |
Natera Horizon™ 421 |
Myriad Foresight® |
Invitae Comprehensive Carrier Screen |
Quest QHerit(R) Extended |
|---|---|---|---|---|---|---|
ABCA3 |
✘ |
✔ |
✘ |
✘ |
✘ |
✘ |
ABCC8 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
ABCD1 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
ACADM |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
ACADVL |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
ACAT1 |
✔ |
✔ |
✔ |
✘ |
✔ |
✔ |
AFF2 |
✘ |
✘ |
✘ |
✘ |
✘ |
✘ |
AGA |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
AGXT |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
AHI1 |
✔ |
✘ |
✔ |
✘ |
✘ |
✘ |
AIRE |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
ALDOB |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
ALPL |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
ANO10 |
✘ |
✘ |
✘ |
✘ |
✘ |
✘ |
ARSA |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
ARX |
✘ |
✘ |
✔ |
✘ |
✘ |
✘ |
ASL |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
ASPA |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
ATP7b |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
BBS1 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
BBS2 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
BCKDHB |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
BLM |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
BTD |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
CBS |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
CC2D2A |
✘ |
✘ |
✔ |
✘ |
✘ |
✘ |
CCDC88C |
✘ |
✘ |
✘ |
✘ |
✘ |
✘ |
CEP290 |
✔ |
✔ |
✔ |
✘ |
✔ |
✔ |
CFTR |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
CHRNE |
✘ |
✔ |
✔ |
✘ |
✔ |
✘ |
CLCN1 |
✘ |
✘ |
✘ |
✘ |
✘ |
✘ |
CLRN1 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
CNGB3 |
✘ |
✔ |
✔ |
✘ |
✔ |
✘ |
COL7A1 |
✘ |
✔ |
✔ |
✘ |
✔ |
✘ |
CPT2 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
CYP11A1 |
✘ |
✔ |
✘ |
✘ |
✘ |
✘ |
CYP21A2 |
✔ |
✔ |
✔ |
✔ |
✔ |
✘ |
CYP27A1 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
CYP27B1 |
✘ |
✔ |
✔ |
✘ |
✘ |
✘ |
DHCR7 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
DHDDS |
✔ |
✔ |
✔ |
✘ |
✔ |
✔ |
DLD |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
DMD |
✔ |
✔ |
✔ |
✔ |
✔ |
✘ |
DYNC2H1 |
✘ |
✘ |
✘ |
✘ |
✘ |
✘ |
ELP1 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
ERCC2 |
✔ |
✘ |
✔ |
✘ |
✘ |
✘ |
EVC2 |
✘ |
✔ |
✔ |
✔ |
✔ |
✘ |
F8 |
✘ |
✘ |
✘ |
✘ |
✘ |
✘ |
F9 |
✘ |
✔ |
✔ |
✘ |
✔ |
✘ |
FAH |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
FANCC |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
FKRP |
✔ |
✔ |
✔ |
✔ |
✔ |
✘ |
FKTN |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
FMO3 |
✘ |
✘ |
✘ |
✘ |
✘ |
✘ |
FMR1 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
FXN |
✘ |
✘ |
✘ |
✘ |
✘ |
✘ |
G6PC |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
GAA |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
GALT |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
GBA |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
GBE1 |
✔ |
✔ |
✔ |
✘ |
✔ |
✔ |
GJB2 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
GLA |
✔ |
✔ |
✔ |
✔ |
✔ |
✘ |
GNPTAB |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
GRIP1 |
✔ |
✘ |
✘ |
✘ |
✘ |
✘ |
HBA1 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
HBA2 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
HBB |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
HEXA |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
HPS1 |
✔ |
✔ |
✔ |
✘ |
✔ |
✔ |
HPS3 |
✔ |
✔ |
✔ |
✘ |
✔ |
✔ |
IDUA |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
L1CAM |
✔ |
✔ |
✔ |
✘ |
✘ |
✘ |
LRP2 |
✔ |
✘ |
✘ |
✘ |
✘ |
✘ |
MCCC2 |
✔ |
✔ |
✔ |
✘ |
✔ |
✔ |
MCOLN1 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
MCPH1 |
✘ |
✘ |
✘ |
✘ |
✘ |
✘ |
MID1 |
✘ |
✘ |
✘ |
✘ |
✘ |
✘ |
MLC1 |
✔ |
✔ |
✔ |
✔ |
✔ |
✘ |
MMACHC |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
MMUT |
✔ |
✘ |
✘ |
✔ |
✘ |
✘ |
MVK |
✘ |
✘ |
✘ |
✘ |
✘ |
✘ |
NAGA |
✘ |
✘ |
✘ |
✘ |
✘ |
✘ |
NEB |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
NPHS1 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
NR0B1 |
✔ |
✔ |
✔ |
✔ |
✔ |
✘ |
OCA2 |
✘ |
✘ |
✘ |
✘ |
✘ |
✘ |
OTC |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
PAH |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
PCDH15 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
PKHD1 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
PLP1 |
✘ |
✘ |
✘ |
✘ |
✘ |
✘ |
PMM2 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
POLG |
✘ |
✔ |
✔ |
✘ |
✘ |
✘ |
PRF1 |
✔ |
✘ |
✔ |
✘ |
✘ |
✘ |
RARS2 |
✔ |
✔ |
✔ |
✘ |
✔ |
✘ |
RNASEH2B |
✔ |
✘ |
✔ |
✘ |
✘ |
✘ |
RPGR |
✔ |
✘ |
✘ |
✘ |
✘ |
✘ |
RS1 |
✘ |
✔ |
✔ |
✔ |
✔ |
✘ |
SCO2 |
✘ |
✘ |
✘ |
✘ |
✘ |
✘ |
SLC19A3 |
✘ |
✘ |
✘ |
✘ |
✘ |
✘ |
SLC26A2 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
SLC26A4 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
SLC37A4 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
SLC6A8 |
✔ |
✔ |
✔ |
✘ |
✔ |
✘ |
SMN1 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
SMPD1 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
TF |
✘ |
✘ |
✘ |
✘ |
✘ |
✘ |
TMEM216 |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
TNXB |
✘ |
✘ |
✘ |
✘ |
✘ |
✘ |
TYR |
✘ |
✔ |
✘ |
✘ |
✘ |
✘ |
USH2A |
✔ |
✔ |
✔ |
✔ |
✔ |
✔ |
XPC |
✔ |
✔ |
✔ |
✔ |
✔ |
✘ |
Quest Diagnostics QHerit™ Extended
Myriad Foresight® Carrier ScreenUniversal Panel
Invitae Comprehensive Carrier Screen
Labcorp / IntegratedInheritest® 500 PLUS Panel
SEMA4 Expanded Carrier Screen (502 Genes)
Natera Horizon 421 (from printed materials provided by Natera)





